HOME | ABOUT US | MEDIA KIT | CONTACT US | INQUIRE
HOME | ABOUT US | MEDIA KIT | CONTACT US | INQUIRE
January 2022
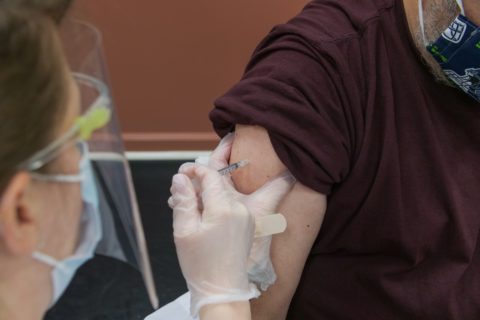 Two measures have been approved by the Centers for Disease Control and Prevention. The first allows increased access to additional doses of Pzifer’s COVID-19 vaccine, the second shortening the recommended interval of time between the initial dose and the booster.
Two measures have been approved by the Centers for Disease Control and Prevention. The first allows increased access to additional doses of Pzifer’s COVID-19 vaccine, the second shortening the recommended interval of time between the initial dose and the booster.
Approval was granted for the two measures on Jan. 4, though just for the Pfizer vaccine. Adjustments were not made for any other COVID-19 vaccine on the market.
Changes to the recommended timing in which individuals receive Pfizer’s version include now being able to receive the booster shot in five months, a deduction from the previously recommended six months.
The agency has not changed the recommended booster interval for people who got other vaccines. The Johnson & Johnson booster interval is two months and the Moderna vaccine can be given six months after initial doses.
The CDC also recommended that kids ages 5 to 11 with moderately or severely weakened immune systems receive an additional dose 28 days after their second Pfizer shot. Currently, only the Pfizer vaccine is recommended for that age group.
The CDC’s decisions followed moves by the Food and Drug Administration on Monday. The FDA also approved Pfizer booster shots for kids ages 12 to 15. The CDC has not announced a recommendation about that, but a CDC expert advisory committee is expected to take up the matter during a meeting Wednesday.